
Vsepr Molecular And Electron Geometry Vsepr Theory Organic Molecules
General Chemistry Map: A Molecular Approach (Tro)

VSEPR Theory — Definition & Overview Expii
Explore molecule shapes by building molecules in 3D! How does molecule shape change with different numbers of bonds and electron pairs? Find out by adding single, double or triple bonds and lone pairs to the central atom. Then, compare the model to real molecules!

Compound molecular geometry table kowerncure
1. Linear: It refers to the geometry shaped by a central atom surrounded by two other atoms. The atoms are arranged in a straight line, and the angle between the bonds is 180 °. The VSEPR notation. Examples of molecules with linear geometry are carbon dioxide (CO 2 ), beryllium chloride (BeCl 2 ), and nitric oxide (NO). 2.
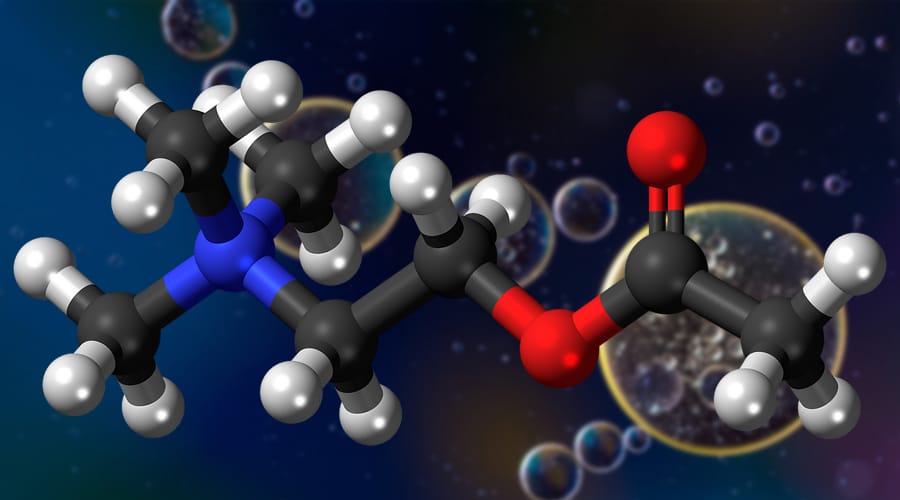
Molecular Geometry Chart Definition, Examples, and Study Guides
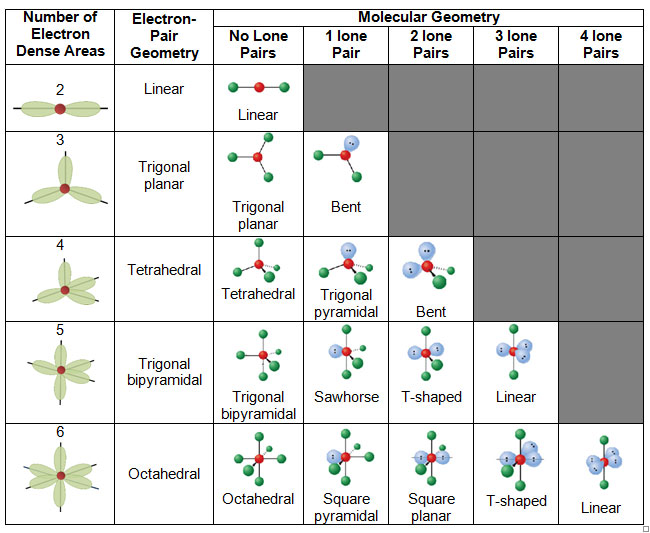
Molecular Geometries The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral. LEARNING OBJECTIVES Apply the VSEPR model to determine the geometry of molecules where the central atom contains one or more lone pairs of electrons. KEY TAKEAWAYS Key Points
Pin by ryan britt on school Molecular geometry, Chemistry classroom
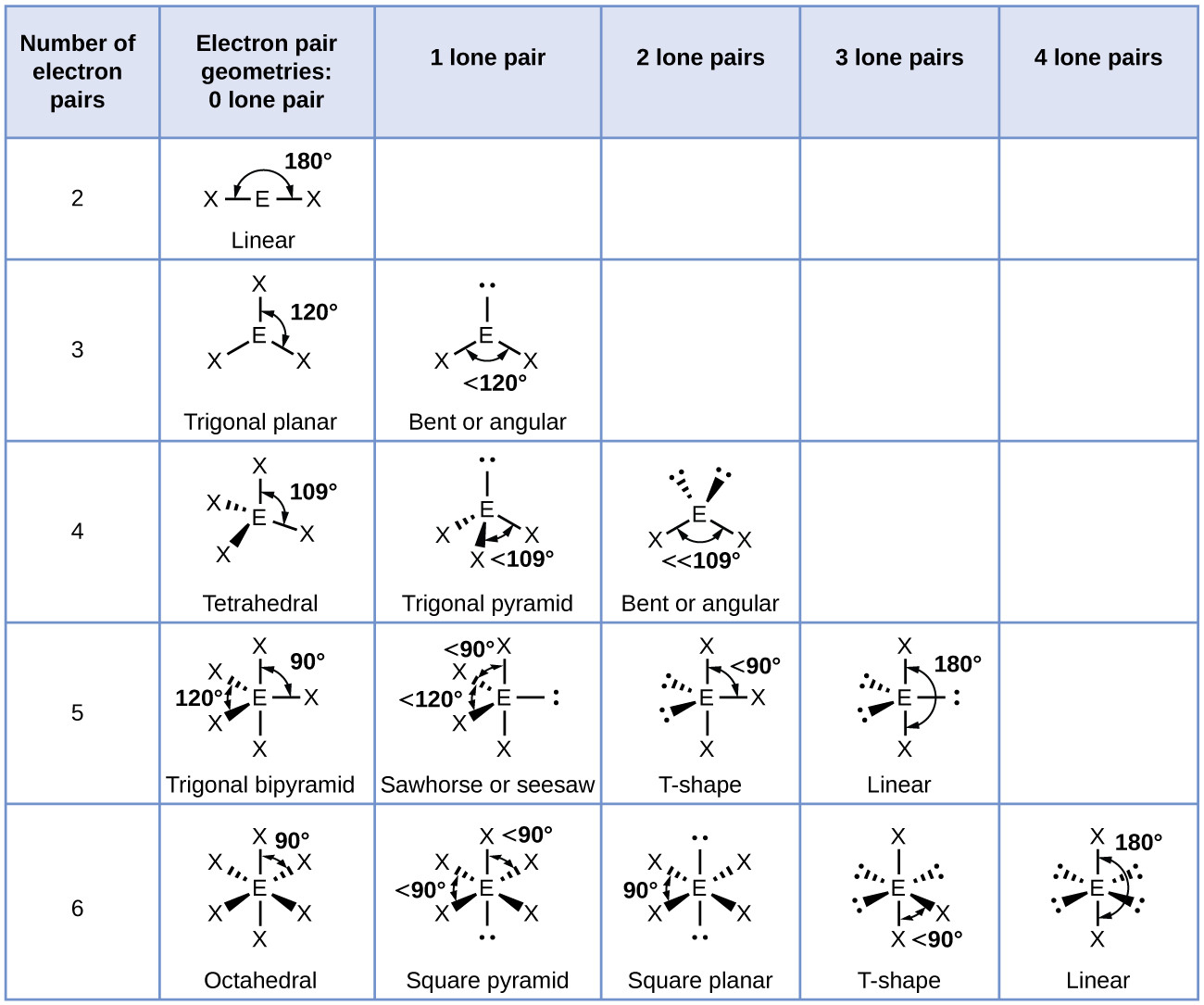
Molecular Geometry Van Koppen/Offen Procedure: draw Lewis Structure, determine Steric Number (SN), Molecular Geometry and Hybridization SN = # of atoms bonded to the central atom plus # of lone pairs on the central atom (SN = the effective number of electron pairs surrounding a central atom).
Lewis Structure And Molecular Geometry Worksheet —
The molecular geometry is bent due to the lone pair of electrons compressing the bond angles to 115.4 o and is represented by AX 2 E. Tetrahedral Electron Pair Geometry and Molecular Shapes. Methane, CH 4, has a tetrahedral electron pair geometry and a tetrahedral molecular geometry, AX 4. The bond angles are 109.5 o. A tetrahedral geometry has.

Electron and Molecular Geometries Chemistry Chemistry, Molecular
VSEPR Theory is short for Valence Shell Electron Pair Repulsion Theory, a method of organizing molecules based on their geometric structures. In chemistry, VSEPR Theory is based on the principle that each atom in a molecule will seek a geometry that maximizes the distance between valence electron pairs, thus minimizing electron-electron.

Pin by Siphora Ketchakeu on organic chemistry 1 Teaching chemistry
Molecular geometry gives information about the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.

moleculargeometrychartmoleculargeometrychart22012653 MOLECULAR
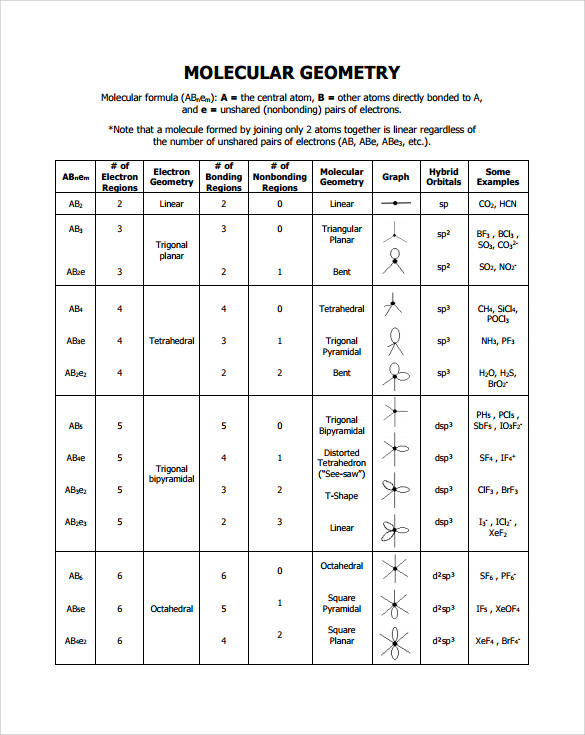
The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms. 2 Linear electron geometry Molecular Geometries VSEPR geometries VSEPR table of molecular geometries

table 1 001.jpg Molecular geometry, Chemistry, Molecular
Basic Molecular Geometries (or Shapes) where the Central Atom has No Lone Pairs Consider a molecule composed of only two types of atoms, A and B: A=central atom B=outer atoms For three or more atoms in a molecule, general formula: AB# (where #=2-6) 3 of outer atoms are at equatorial positions, 120° from each other
FREE 8+ Sample Molecular Geometry Chart Templates in PDF MS Word
Chapter 21 Index By the end of this section, you will be able to: Predict the structures of small molecules using valence shell electron pair repulsion (VSEPR) theory Explain the concepts of polar covalent bonds and molecular polarity Assess the polarity of a molecule based on its bonding and structure

amppinterest in action Molecular geometry, Molecular, Study tools
Molecular geometry is the three-dimensional arrangement of the atoms that constitute a molecule. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.
Untitled Document
1. Linear: It specifies the geometry shaped by a central atom surrounded by two other atoms. The atoms are arranged in a straight line, and the angle between the bonds, or bond angle, is 180 °. The VSEPR notation is AX 2. Examples of molecules with linear geometry are carbon dioxide (CO 2 ), beryllium chloride (BeCl 2 ), and nitric oxide (NO). 2.

Different Shapes CHEMISTRY COMMUNITY
Valence shell electron pair repulsion or VSEPR theory can be used to predict molecular geometry. The theory is based on Lewis structures and the simple idea that that the preferred geometry around a central atom is the one that minimizes electron repulsion.. She considers chemistry to be a bridge between biology and physics that can help.

Pin on IB Chemistry
Ketzbook explains molecular geometry, VSEPR theory, and the 5 basic shapes of molecules with examples for each one.For a limited time, earn double FREE stock.

Electron Geometry VS Molecular Geometry Difference between Electron
Figure 8.6.1 8.6. 1 shows the various molecular geometries for the five VESPR electronic geometries with 2 to 6 electron domains. When there are no lone pairs the molecular geometry is the electron (VESPR) geometry. When there are lone pairs, you need to look at the structure and recognize the names and bond angles.